Phosphosignaling networks are complex. Bacteria transmit signals through reversible phosphorylation using the classical two component systems, and recent genomic data, in particular from the Ocean Genome Sampling Initiative, have shown that bacteria also rely on Ser/Thr kinases for cell signaling. A case in point is Mtb, which codes for the same number of two component systems as Ser/Thr kinases. These systems are less well understood, and our view of Ser/Thr signaling is largely shaped by what is known in the equivalent eukaryotic systems. However, bacteria very likely evolved their own components and mechanisms to signal through phosphate. We are exploring these possibilities by using unbiased, systems-wide approaches to uncover new, uniquely bacterial themes in Mtb phosphorylation and its cellular effects. We also recently identified extensive phosphorylation on Tyr in Mtb, suggesting that this modification is more common in bacteria than previously thought.
References
Agents of change – concepts in Mycobacterium tuberculosis Ser/Thr/Tyr phosphosignalling. Sherman DR, Grundner C. Mol Microbiol. 2014 Oct;94(2):231-41. doi: 10.1111/mmi.12747. Epub 2014 Aug 25. Review.
Mycobacterium tuberculosis supports protein tyrosine phosphorylation. Kusebauch U, Ortega C, Ollodart A, Rogers RS, Sherman DR, Moritz RL, Grundner C. Proc Natl Acad Sci U S A. 2014 Jun 24;111(25):9265-70. doi: 10.1073/pnas.1323894111. Epub 2014 Jun 9.
Mycobacterium tuberculosis Ser/Thr protein kinase B mediates an oxygen-dependent replication switch. Ortega C, Liao R, Anderson LN, Rustad T, Ollodart AR, Wright AT, Sherman DR, Grundner C. PLoS Biol. 2014 Jan;12(1):e1001746. doi: 10.1371/journal.pbio.1001746. Epub 2014 Jan 7.
Osmosensory signaling in Mycobacterium tuberculosis mediated by a eukaryotic-like Ser/Thr protein kinase. Hatzios SK, Baer CE, Rustad TR, Siegrist MS, Pang JM, Ortega C, Alber T, Grundner C, Sherman DR, Bertozzi CR. Proc Natl Acad Sci U S A. 2013 Dec 24;110(52):E5069-77. doi: 10.1073/pnas.1321205110. Epub 2013 Dec 5.
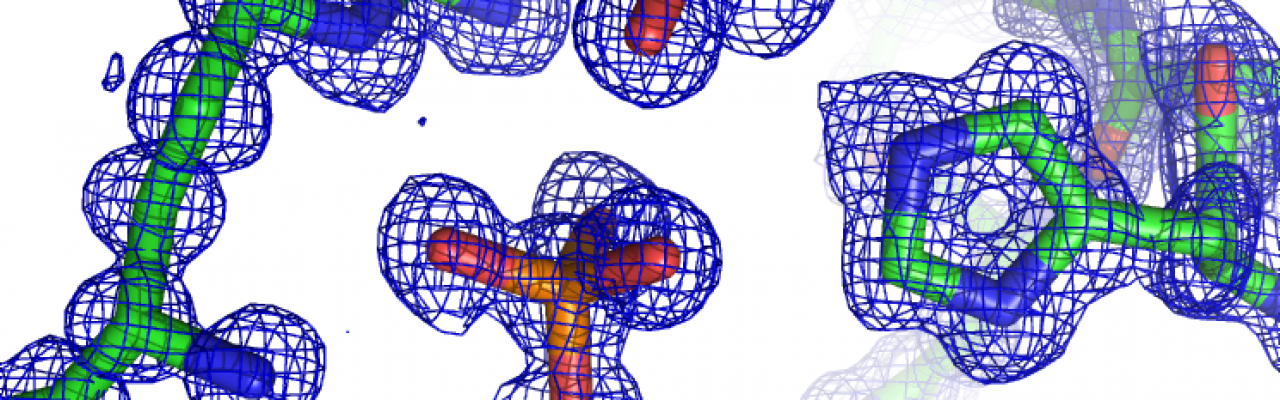
More than 30% of all Mtb genes are hypothetical or unknown. This lack of annotation is a bottleneck for studying Mtb on every level. Discovering the function of unknown proteins is difficult, particularly for species-specific proteins, for which sequence-base d methods of annotation fail. We are using high-throughput experimental tools to identify biochemical functions of proteins. By using chemical probes that detect activity coupled to mass spectrometric identification of target proteins, we survey the proteome for unknown activities. We provided the most comprehensive description of the ATPase family in Mtb, a large and central protein family. Combined with structural biology, we discover new protein families and explore even the most divergent sequence space. Some microbial pathogens pose even bigger annotation challenges: Less than half of all proteins in Plasmodium falciparum, for example, have known functions. We are now extending our chemical proteomic platform to also annotate unknown Pf proteins and to discover enzymes that control life cycle decisions.
References
Crystal structures of Mycobacterial MeaB and MMAA-like GTPases. Edwards TE, Baugh L, Bullen J, Baydo RO, Witte P, Thompkins K, Phan IQ, Abendroth J, Clifton MC, Sankaran B, Van Voorhis WC, Myler PJ, Staker BL, Grundner C, Lorimer DD. J Struct Funct Genomics. 2015 Jun;16(2):91-9. doi: 10.1007/s10969-015-9197-2. Epub 2015 Apr 2.
Mycobacterium tuberculosis Rv2179c protein establishes a new exoribonuclease family with broad phylogenetic distribution. Abendroth J, Ollodart A, Andrews ES, Myler PJ, Staker BL, Edwards TE, Arcus VL, Grundner C. J Biol Chem. 2014 Jan 24;289(4):2139-47. doi: 10.1074/jbc.M113.525683. Epub 2013 Dec 4.
Identification of widespread adenosine nucleotide binding in Mycobacterium tuberculosis. Ansong C, Ortega C, Payne SH, Haft DH, Chauvignè-Hines LM, Lewis MP, Ollodart AR, Purvine SO, Shukla AK, Fortuin S, Smith RD, Adkins JN, Grundner C, Wright AT. Chem Biol. 2013 Jan 24;20(1):123-33. doi: 10.1016/j.chembiol.2012.11.008.
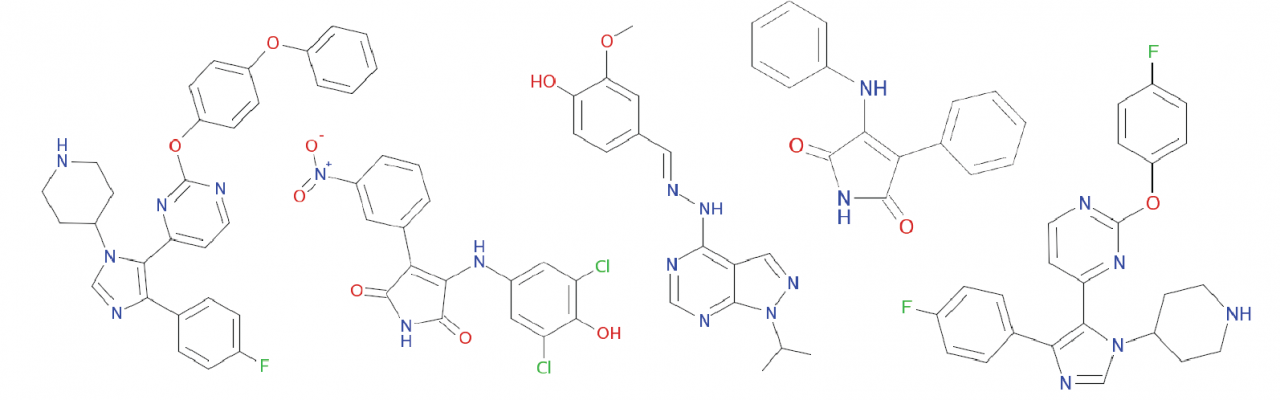
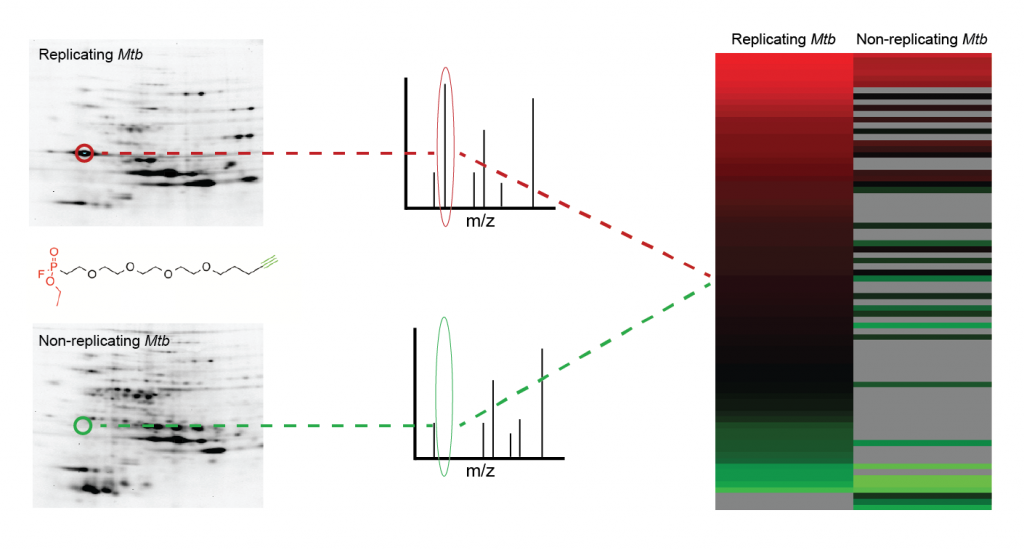
Mtb causes a chronic, persistent infection that is due to slow or non-replicating bacteria. These non-replicating bacteria are drug tolerant, requiring treatment of at least 6 months. To shorten treatment times and to avoid the emergence of heritable drug resistance, targeting persistent Mtb is a priority. However, as persisters are metabolically inactive, most drugs lose their efficacy against their targets in these non-replicating states. We use chemical proteomics to discover the physiologic pathways that are required for maintaining persistence and that remain vulnerable for therapeutic targeting during non-replication. Other organisms rely on related transitions from replication to non-replication to facilitate infection or transmission. Plasmodium falciparum, for example, develops from a replicating schizont to a non-replicating gametocyte that is poised for transmission to the insect vector. Using our chemical proteomics approach, we measure enzyme activities associated with this developmental switch and define drug targets that can interfere with Pf transmission.
References
Systematic Survey of Serine Hydrolase Activity in Mycobacterium tuberculosis Defines Changes Associated with Persistence. Ortega C, Anderson LN, Frando A, Sadler NC, Brown RW, Smith RD, Wright AT, Grundner C. Cell Chem Biol. 2016 Feb 18;23(2):290-8. doi: 10.1016/j.chembiol.2016.01.003. Epub 2016 Feb 4.
Mycobacterium tuberculosis Ser/Thr protein kinase B mediates an oxygen-dependent replication switch. Ortega C, Liao R, Anderson LN, Rustad T, Ollodart AR, Wright AT, Sherman DR, Grundner C. PLoS Biol. 2014 Jan;12(1):e1001746. doi: 10.1371/journal.pbio.1001746. Epub 2014 Jan 7.